Portable electrical safety tester
SECULIFE ST Series
The portable electrical safety tester SECULIFE ST series offers antibacterial properties and complies with standard testing procedures. It is designed for testing biotech medical equipment, electrical products, post-repair testing, and more (IEC 62353 / EN 62353 (VDE 0751), IEC 62638, IEC 60601, DIN VDE 0701-0702, DGUV regulation 3, MPBetreibV).
Its testing functions include ground resistance testing, high voltage testing, insulation resistance testing, leakage current testing, and function testing (Depend on model).
User-Friendly Interface Design
- Antibacterial-certified housing
- Supports single-step tests and sequential testing procedures
- Editable test procedures and reports
- Preloaded with various test procedures for quick initiation of standard tests
- Intuitive interface with a color touchscreen, large rotary knob, and function keys
- Compatible with laser label printers for direct report printing
- Optional adapters for applied parts from major medical equipment brands (contact for details)
- supports the dedicated biomedical testing software “IZYTRONIQ.”
Comprehensive Measurement Functions and Data Storage
- Simultaneous measurement of multiple data points for convenient record-keeping
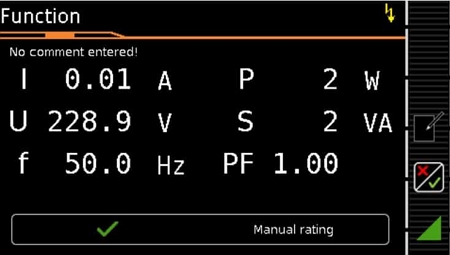
- Functional testing capabilities: Voltage (V), Current (I), Power (W), Frequency (F), Power Factor (PF)
- Equipped with 10 applied parts for measurements
- New voltage measurement input, allowing use as a multimeter for direct voltage measurements
- Automatic pre-test detection of connections and protection of the device under test
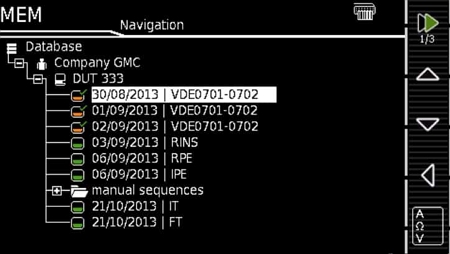
- Powerful database system capable of storing up to 50,000 records
- USB-A and USB host ports for data access and PC connectivity
- Multilingual support available (including keyboard, character sets, date, and time)
/div>
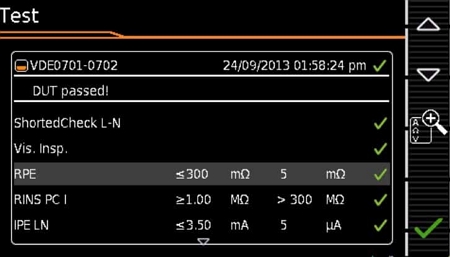
Supports Key Electrical Safety Testing
- Hipot test
- 500V Insulation Resistance Testing
- 25A Ground Continuity Testing
- Supports various PRCD tests such as PRCD-S/PRCD-K and ground continuity switching, integrated into the “VDE 0701-0702-PRCD” testing program
- Leakage current testing for single-phase and three-phase input devices (optional)
- Leakage current tests in compliance with medical equipment standards, including protective earth leakage current and patient leakage current
/div>
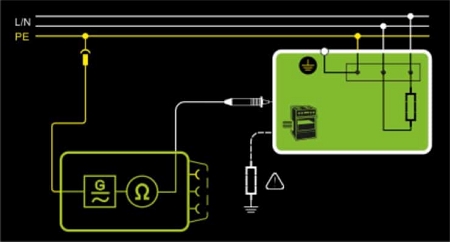
Compliance with Standard Testing Requirements
- IEC 62353 / EN 62353 (VDE 0751)
- DIN VDE 0701-0702 (including PRCDs)
- IEC/EN 60974-4 (VDE 0544-4)
- IEC 60601
- IEC 61010
Built-in SECUTEST DB+ database expansion:
- Direct control via PC software (IZYTRONIQ)
- Additional database fields: property, building, floor, and room for clearer data structuring
- Instrument department and cost center integration
- Batch printing of multiple reports (Z721S thermal printer) with real-time printing support
- Customizable report formats stored in the SECULIFE ST PRO, including company logos
- Export test data to USB drives
- Import test procedures from PC software (IZYTRONIQ) or USB drives
- PC software (IZYTRONIQ) allows creation and upload of custom test procedures
SECULIFE ST PRO Built-in SECUTEST DB COMFORT Database Expansion
- Latest “Medical Test Database”
- Database search configured for “UDI” (Unique Device Identification)
- Expanded to 24 customizable test procedures
- Touchscreen editing for direct on-screen modifications
- Auto-save function for automatic test result storage
- Test data can be directly saved to a PC without requiring transfer through the ST PRO



 /div>
/div> /div>
/div>